 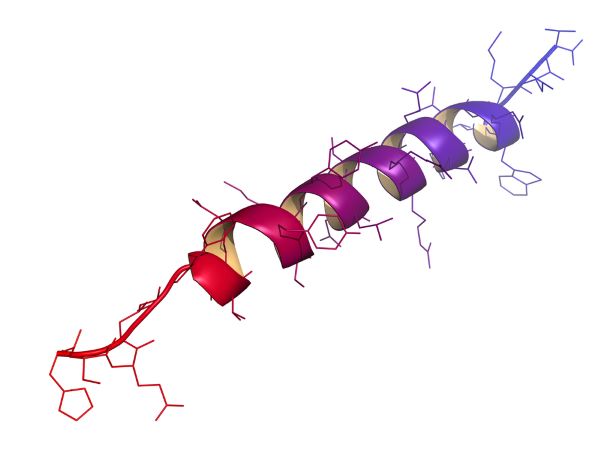
Pauling's remarkable insights led to the correct formulation of the hydrogen-bonded helical structures of polypeptides ( Pauling et al. An interesting feature of the Bragg, Kendrew and Perutz paper is the consideration of intramolecularly hydrogen-bonded structures, which were later shown to be stereochemically and energetically unacceptable. This expectation has been amply fulfilled in the last half century with the result that almost every feature of polypeptide chain folding is now firmly established by experimental studies. (1950) in a paper that concluded that ‘there appears a real simplicity of chain structures in myoglobin, which will perhaps be shown by other favourably built proteins… There is hope that the study of such proteins may lead to a reliable determination of structure’. Attempts to rationalize limited X-ray diffraction data in terms of regular hydrogen-bonded polypeptide structures were summarized by Bragg et al. The construction of globular polypeptide structures using diverse hybrid sequences appears to be a realistic possibility.Ī little over 60 years ago, Maurice Huggins reviewed the hydrogen-bonded structures of polypeptide chains, at a time when no detailed structural information was available on oligopeptides and proteins ( Huggins 1942, 1943). The achiral γ-residue gabapentin is a readily accessible building block in the design of peptides containing γ-amino acids. This approach is exemplified by well-folded structures like the C 12 (αγ) and C 14 (γγ) helices formed in short peptides containing multiply substituted γ-residues. 
The use of stereochemically constrained ω-residues promises to expand the range of peptide design strategies to include ω-amino acids. The present review explores the relationships between folded conformations in hybrid sequences and their counterparts in all α-residue sequences. Examples of both peptide helices and hairpins are presented. Several recent examples demonstrate that individual ω-amino acid residues and hybrid dipeptide segments may be incorporated into the regular structures of α-peptides. 
The available crystallographic information on hydrogen-bonded hybrid turns is surveyed in this review. Hybrid turns may be classified by comparison with their αα counterparts. 
The β-turn, a ubiquitous structural feature formed by two residue (αα) segments in proteins and peptides, is stabilized by a 10-atom (C 10) intramolecular 4→1 hydrogen bond. This review examines the nature of intramolecularly hydrogen-bonded conformations of hybrid peptides formed by amino acid residues, with a varying number of backbone atoms. More recently, the discovery of well-folded structures in oligopeptides containing β-amino acids has focused a great deal of current interest on the conformational properties of peptides constructed from higher homologues (ω) of α-amino acids. An extraordinary wealth of conformational information is now available on peptides and proteins, which are formed of α-amino acid residues. Half a century has passed since the hydrogen-bonded secondary structures of polypeptides and proteins were first recognized. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
